Last Updated on October 22, 2023 by Kevin Chen
Batteries are designed to provide the power needed to run various devices, allowing you to take your favorite gadgets with you. Consumers today have access to a wide range of battery options, including nickel-cadmium batteries.
What is Nickel-Cadmium Battery
A rechargeable nickel-cadmium battery (NiCad or NiCad) is used in portable computers, drills, camcorders, and other small battery-operated equipment requiring even power discharge. Nickel oxide hydroxide electrodes, metallic cadmium electrodes, and a potassium hydroxide alkaline electrolyte are used in NiCds. Waldemar Junger created and patented the NiCad battery in 1899.
Types of Nickel Cadmium Battery Pack
Nickel Cadmium battery packs come in thousands of distinct configurations. There are several battery cell configurations, voltages, amperages (amps), and various combinations of all three. Individual nicad cells have been wired, welded together, glued, and packaged in shrink-wrap with a unique part. Nickel Cadmium battery packs usually are made up of a combination of individual nicad cells that have been wired, welded together, bonded, and packed in shrink-wrap with a unique part.
These nicad cells can be stacked vertically, in two even rows, in two uneven rows, in horizontal rows, in two horizontal rows, in two nested horizontal rows, in a triangle, and in various forms during manufacture. The several nicad assembly combinations are necessary to fit into certain items with limited space and provide the appropriate voltage and amp-hours required by the product.
Features of Ni-Cd batteries
In 1899, a Swede named Waldemar Jungner invented nickel cadmium (Ni-Cd or “nicad”) batteries. Porous electrodes were utilized to contain nickel, which was fashioned into pockets for a nickel-cadmium combination to sit in and conduct electricity.
Smart individuals discovered that the larger the surface area within a battery, the better for stronger electric currents after decades of research and trial. As a result, Ni-Cd batteries now have a porous anode, and cathode plates sandwiched between a separator and rolled up into a delectable charge with maximum power output.
An electrode is a spot on either end of a Ni-Cd battery where electricity enters and exits. The positive terminal of Ni-Cd batteries contains nickel hydroxide, while the negative terminal contains metallic cadmium. A liquid alkaline electrolyte solution, commonly potassium hydroxide, is contained therein.
Cadmium is a highly poisonous metal that must be disposed of correctly. You can take them to a specific battery recycling site in the United States. Even though current Ni-Cd batteries effectively retain cadmium within the battery and do not leak, the European Union banned portable Ni-Cd battery types in 2008.
What distinguishes Ni-Cd rechargeable batteries from traditional alkaline batteries
Ni-Cd cells come in the exact sizes as alkaline cells, from AAA to sub C and D, and multi-cell combo packs containing the equivalent of a 9-volt battery.
Other alkaline batteries can pump out similar amps and have similar milliamp-hours (mah) of capacity. They’ve been employed in various applications over the years, including home portable phones, vehicular toys, flashlights, power tools, and picture equipment. Ni-Cd batteries differ from traditional alkaline batteries in various ways, including:
They are rechargeable
Ni-Cd batteries, for example, may be recharged, but most alkaline batteries must be discarded once they’ve been used up.
Place them in a battery charger for a charge cycle, and they’ll be ready to use again. They can be destroyed if overcharged, so it’s best to remove them from the battery charger once they’ve reached 100 percent charge.
Constant current
Most alkaline batteries have a cell voltage of around 2V, whereas Ni-Cd batteries have a cell voltage of 1.2V.
While this may appear to be a disadvantage, Ni-Cd batteries can produce 1.2 volts at full power from each battery cell until the battery is entirely depleted. While other batteries gradually lose power during a charge, Ni-Cd batteries maintain a consistent voltage throughout their cycle life.
You may have had the experience of using a power tool like a drill that, with a new charge, screws the daylights out of anything it comes into contact with. After 10 to 12 applications, a power differential is obvious, and the drill emits a depressing sluggish whirr before grinding to a standstill.
You may have had the experience of utilizing a power tool, such as a drill, that screws the living daylights out of anything it comes into contact with after a full charge. The drill then emits a dismal sluggish whirr and grinds to a halt after 10 to 12 operations, at which point a power difference becomes apparent.
Fast Charging and Discharging
Ni-Cd batteries’ low internal resistance allows them to quickly discharge and charge a large amount of power.
Can perform well in temperature extremes
Ni-Cd batteries do not quickly overheat due to their low resistance. As a result, they may be used in various harsh situations with a wide range of temperatures.
Longevity
Ni-Cd batteries have a long life span. You can expect them to work for years as long as they are not overcharged. Too much charge can damage the battery’s vent, causing the cells to dry up.
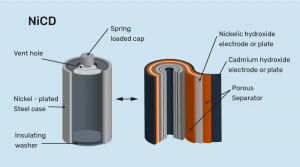
Construction of NiCad Batteries
The NiCad battery’s basic unit is the cell. All included are positive and negative plates, separators, electrolytes, cell vents, and cell containers. The positive plates are manufactured from a porous plaque that has been coated with nickel-hydroxide. The negative plates are created from cadmium-hydroxide-deposited plaques similar to the positive plates. In both cases, the porous plaque is made by sintering nickel powder onto a fine-mesh wire screen.
Sintering is a high-temperature technique that fuses extremely minute powder grains. The plaque is created and sliced into the right plate-size after the active positive and negative elements are placed. After that, each plate’s corner is welded with a nickel tab. Moreover, the plates are linked by soldering the tabs to the proper terminals. A continuous strip of porous plastic separates the plates from one another.
The NiCad battery’s electrolyte is a 30 percent potassium hydroxide (KOH) solution in distilled water. At room temperature, the electrolyte’s specific gravity remains between 1.240 and 1.300. It’s worth noting that the electrolyte doesn’t vary much during charge or discharge. As a result, a specific gravity check of the electrolyte cannot be used to assess the battery charge. The electrolyte level should be maintained above the tops of the plates.
Charging NiCad batteries
When a NiCad battery receives a charging current, the negative plates lose oxygen and begin to create metallic cadmium. The positive plates’ active substance, nickel-hydroxide, become more oxidized. This process continues until all of the oxygen in the negative plates has been removed, leaving only cadmium. The cells release gas near the end of the charging cycle. If the cells are overcharged, this will also happen. The electrolyte’s water decomposes into hydrogen at the negative plates and oxygen at the positive plates, resulting in this gas. When gassing occurs, it is determined by the voltage used while charging and the temperature. Some gassing, however minor, is required to charge a NiCad battery fully; consequently, water will be employed.
Discharge
During discharge, the chemical action is reversed. The positive plates gradually lose oxygen, which the negative plates reclaim. As a result of this process, chemical energy is transformed to electrical energy. The plates absorb the electrolyte during discharge. The electrolyte level grows when the battery is recharged, and at full charge, the electrolyte level is at its highest. As a result, water should only be introduced once the battery has been fully charged.
Changing from lead-acid to NiCad
Lead-acid batteries and NiCad batteries are frequently interchangeable. The battery compartment must be clean, dry, and clear of all traces of acid from the previous battery when replacing a lead-acid battery with a NiCad battery. The compartment must be cleaned appropriately and neutralized with ammonia or boric acid solution before coating with an alkali-resistant varnish.
Before connecting the battery vent system, the pad in the battery sump jar should be saturated with a 3 percent (by weight) solution of boric acid and water.
Servicing NiCad batteries
There are considerable differences between the NiCad and lead-acid battery servicing methods. The following are the most crucial things to keep in mind.
NiCad batteries should be kept in a separate storage and maintenance room. Chemically, the electrolyte is the polar opposite of sulphuric acid, which is utilized in lead-acid batteries. The electrolyte in a nickel-cadmium battery can be contaminated by fumes from a lead-acid battery. This precaution should apply to lead-acid battery-related devices such as hand tools and syringes. Indeed, everything feasible must be done to keep acid-containing items out of NiCad battery stores.
NiCad batteries’ potassium hydroxide electrolyte is particularly corrosive. When handling and repairing batteries, safety equipment such as goggles, rubber gloves, and rubber aprons should be worn. Appropriate washing facilities must be available if the electrolyte is spilled on clothing or the skin. Any electrolyte exposure should be washed with water, vinegar, lemon juice, or a boric acid solution. When mixing potassium hydroxide and distilled water to generate an electrolyte, keep in mind that the potassium hydroxide should be gently added to the water, not the other way around.
When cleaning the battery, avoid using a wire brush. If a wire brush is used, severe arcing may occur. Additionally, the vent ports should be closed during the cleaning procedure, and the battery should never be cleaned with acids, solvents, or any chemical solution. Potassium carbonate crystals can occur when an electrolyte is spilled and reacts with carbon dioxide. These can be loosened with a fiber brush and washed off with a moist towel because they are non-toxic and non-corrosive. When potassium carbonate builds on a fully serviced battery, it could signal that the battery is overcharging due to an out-of-adjusted voltage regulator.
Water should never be added to the battery until it has been fully charged three to four hours after it has been fully charged. Only use distilled or demineralized water if you need to add water. Also, take care not to overcharge the battery with water. If you do this and have to remove part of the liquid, the concentration of potassium hydroxide in the cell will be reduced. This will have an impact on how it works.
The specific gravity of the electrolyte does not vary significantly since it does not react chemically with the cell plates. As a result, a hydrometer cannot be used to determine the level of charge of a NiCad battery. Furthermore, a voltage test cannot detect the charge of a NiCad battery because the voltage of a NiCad battery remains constant for 90% of the discharge cycle.
Servicing intervals of NiCad
Because water consumption varies with ambient temperature and operation methods, NiCad batteries should be serviced at regular intervals depending on experience. The battery should be taken from the aircraft and checked regularly in the shop.
Some cells in a battery can achieve zero potential and charge in the opposite direction if it is entirely depleted. This could affect it, causing it to lose its full capacity charge. If this occurs, the battery should be emptied and each cell balanced before recharging. This is referred to as equalization.
The constant-voltage or constant-current methods of charging are also viable options. Maintain the charging voltage constant until the charging current decays to 3 amperes or less for constant potential charging, ensuring that the battery cell temperature does not reach 100 degrees Fahrenheit and the voltage begins to fall.
Trickle charging
Trickle charging repeatedly charges a battery in an overcharge condition to keep it in standby mode. Even though some manufacturers do not encourage this charging technique, some operators have selected it for their NiCad batteries. Keep in mind that, due to the gassing effect mentioned previously, utilizing a trickle charger will deplete water over time. Before putting the battery on the plane, ensure the electrolyte level is correct. If this is not done, there is a risk of a battery incident since the cells may dry out before the maintenance time is completed.
Storage Recommendations
A Nickel-Cadmium battery can be stored in either a charged or discharged state. On the other hand, long-term storage might hasten battery drain and result in reactant deactivation. Even though the cells may be stored at temperatures ranging from -20°C to +45°C, high temperatures can cause the chemicals to crumble, so it’s best to keep them in a cold, non-corrosive, clean, and dry environment. It may take two or three deep discharge cycles to recover the battery’s full capacity if it has been kept perfect for an extended period.
Conclusion
When charged with a dynamo, NiCad batteries heat up excessively, enter thermal runaway mode, and self-destruct – even in over-current shutdown systems. However, most NiCad battery packs have an inside thermal charger cutoff that signals when the battery heats up and achieves maximum voltage.
Lastly, we at ICRFQ are the best electrical components manufacturer in China. If you need any electrical component contact us, we manufacture quality and affordable components that will serve their purpose without need for worry or doubt.
If you want to find more Electronic Components Distributors, please check out the following articles:
Electronic Components Distributors In the USA
Electronic Components Distributors In UK
Electronic Components Distributors In China
Electronic Components Distributors In India
Electronic Components Distributors In Singapore
Electronic Components Distributors In Malaysia
Electronic Components Distributors In Vietnam
Electronic Components Distributors In South Korea
- Where to buy IC chips? The Best Guide? - March 26, 2024
- Breaking Down Barriers: Overcoming Obstacles in Cross-Border Electronic Component Trade - March 4, 2024
- Everything You Need to Know About Amplifier IC Chips - March 4, 2024